
:C O : Given that oxygen is divalent and carbon is tetravalent. (We will discuss electronegativity in more detail in Unit 2 the electronegativity trend for second row elements are: Li < Be < B < C < N < O < F, with F being most electronegative. The formal charge of carbon monoxide can be determined for the Lewis structure given below. Lewis structures with negative formal charges on more electronegative atoms are preferable.Lets find the formal charge for the molecule from the question. 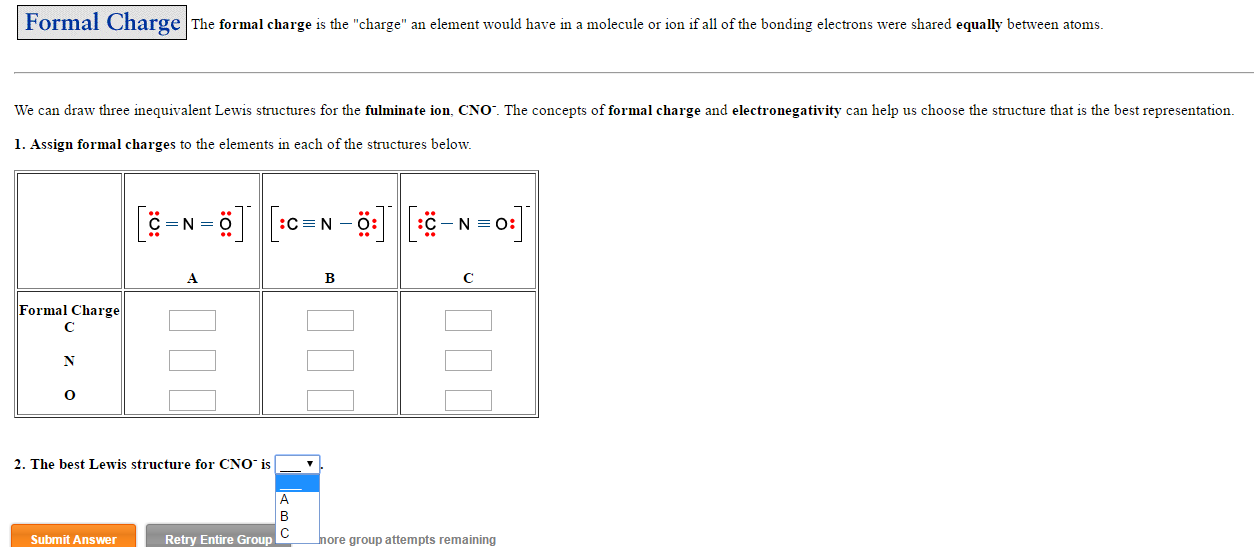
A Lewis structure with formal charges of the same sign (both + or both −) on adjacent atoms is less likely. Formal charge formula: Formal charge valence electrons - nonbonding electrons - (.The smaller the magnitude of the formal charges, the better. To calculate the total outermost electrons of a particular element, multiply the number of atoms present in that element by its valence shell electrons.The fewer atoms with nonzero formal charges, the better.For an uncharged molecule, a Lewis structure in which all atoms have a formal charge of zero is preferable.Therefore, if following the steps for drawing Lewis structures leads to more than one possible arrangement of electrons and/or atoms for a given molecule, formal charges can help to decide which arrangement is likely to be the most stable, and hence the most likely Lewis structure for the given molecule. or q), in the covalent view of chemical bonding, is the hypothetical charge assigned to an atom in a molecule. While formal charges do not portray the true electron density distribution within a molecule, they nonetheless account for electron arrangement in a Lewis structure in units of whole electron. Using Formal Charge to Predict the Most Likely Lewis Structure This lecture is about how to calculate formal charge on an atom in a molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
